
Pharmaceutical Industry

properQMR
Our original, namesake solution, for the pharmaceutical industry
Quality Management Review (QMR) in the pharmaceutical manufacturing industry is an obligatory, regulated, managerial process designed to ensure oversight while concurrently creating a platform for feedback, regulatory/audit transparency, and process improvement. properQMR offers a consultative, software-supported approach for managing QMR, and transforms the requisite data collection process into an ongoing series of opportunities for cross-organizational improvement.
Data entry, calculations of derived parameters (ex. KPIs), detailed analysis, and presentation in graphical and contextual format are delivered in one useable format:
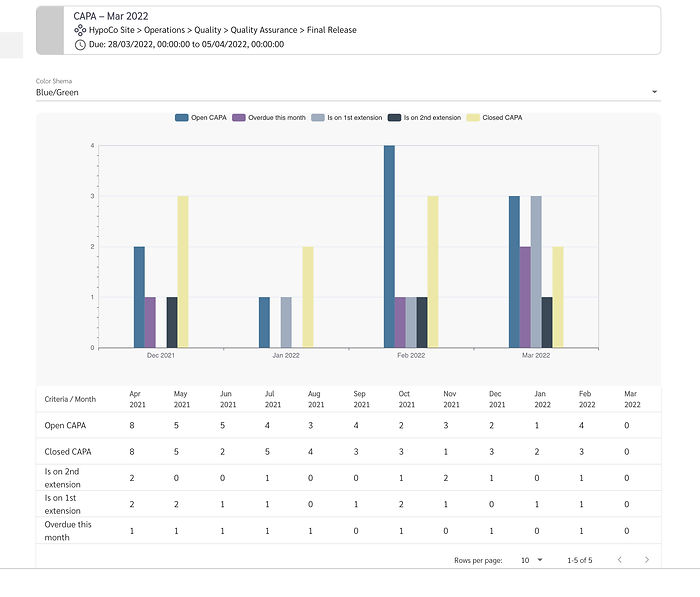
Our solution is a combination of consultant-guided data analysis and a customisable software platform. The properQMR solution allows users to analyse trends in their data which can be visualised, aggregated, and differentiated by unit, department, geographical location, etc. At the same time, end-to-end encrypted data is available to management in real time and can be analysed on any desktop or mobile device:

Finally, any interaction with the software is stored in a revision-proof audit facility, providing maximum transparency and security for management, auditors, and regulators.
Based on ICH Q10, Pharmaceutical Quality System, management is directed to regularly review the performance of all relevant quality systems (processes). This is, even in 2022, generally implemented using handwritten documents or spreadsheets which are, in turn, presented to management during monthly QMR meetings as pure data. Since the approach is document driven, a detailed analysis of data is close to impossible, and an opportunity to analyse the data, on any number of levels, has been lost.
Though the formal requirements of a QMR process are being met, the intended, continuous improvement, based on transparent, data-based analysis does not take place, leaving the QMR process a dull and time-consuming task.
The properQMR approach is a 5-phase process, with clearly defined, independent and scalable modules allowing deployment in any size organisation:
Phase 1. Data gathering/collection: A small team of specialists performs an initial on-site data analysis. Depending on the requisite scope of the data this process will occur over a one-to-two-week period
Result: Data points to be monitored, configuration of ProperQMR software.

Phase 2. Configure ProperQMR Software. Based on the Phase 1 analysis, the team, together with head office programmers, configures & customises the software. This task takes two to three weeks, depending on the Phase 1 findings.
Result: Ready-for-deployment, customised software package/suite/etc.
Phase 3. Software deployment and validation. Define Access Control Lists (ACLs), roles and users for software on-/off-site. Installation of software as Virtual Machine (VM) on local VM host, “properQMR Machine”, or cloud-based platform depending on client preference. Connectivity to existing software to enable automated data gathering. Creation of interfaces and implementation of interfaces at the client's facility. This phase requires approximately one to three weeks.
Result: Ready-to-use in-house or cloud-based properQMR software, ongoing support by software development/training team.
Phase 4. Training. Online, virtual training in properQMR software, supported by a dedicated training specialist. Training includes QMR process and properQMR software training.
Result: Users ready to use properQMR software and execute the QMR processes.
Phase 5. Ongoing feedback and improvement. Guided by our highly experienced/Industry expert consultants, results are re-factored, and companywide processes can be continuously analysed and improved based on internal or cooperative properQMR analysis. Driven by client-specific process optimisation requirements, we may support review and analysis, as well as assist in QMS process improvement with the objective of e.g., reduction of negative parameter occurrences (quality processes).
Result: Observations made in the QMR meetings can be turned into requests for process optimisation and improvement. ProperQMR support in the execution of tangible process improvements through data supported analysis of current state and identification of potential in client-established processes.
